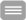Research Group Neuroglia
Projects
To evaluate the role of glia in neurodegenerative diseases and to investigate putative glia-mediated neuroprotective strategies. Our group is currently working on the following aspects:
1. Evaluation of glial-mediated inflammatory processes in the retina after acute retinal ischemia. Effect of ageing and modulation by hydroxy-methyl-glutaryl CoA reductase inhibitors (statins)
Retinal injury and retinal disease lead to neural cell death and are therefore correlate with inflammation. Dependening on the severity of the injury, immunocompetent cells within the retina, retinal microglia, become activated, however, peripheral blood cells which can cross the damaged blood-retinal barrier are also recruited. Both microglia and macrophages phagozytose neuronal debris and can have both beneficial (promote regeneration) and also harmful (promote cell death) effects on the damaged tissue but depending on the severity of the lesion and also possibly on the age of the animal.
Herein we analyse the cellular response of retinal microglia and that of invading macrophages, as well as the expression pattern of pro- and anti-inflammatory cytokines after transient retinal ischemia in young adult and aged rodents. The putative effect of statin treatment on inflammation after injury is also evaluated.
2. Analysis of injury-mediated cell response of endogenous stem- and progenitor-like cells from the rodent retina. Retinal microglia as putative progenitor-like cells of the CNS
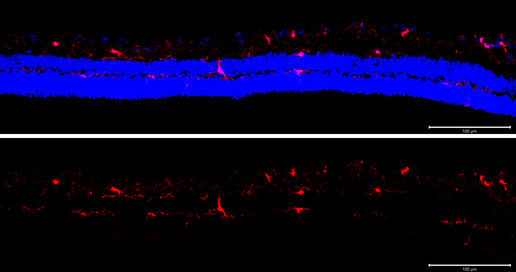
During the last decade, retinal stem-/progenitor-like cells of different ocular regions of the adult mammalian eye have been identified in vitro, including epithelial cells of the ciliary body and Müller glia of the neural retina. Interestingly, Müller glia are capable of transdifferentiat into neurons, in particular after exogenous stimulation (injury or growth factor delivery). In this study, we analyse whether this “dormant” neurogenic potential of residing adult progenitor-like ocular cells can be induced by experimental injury in vivo.
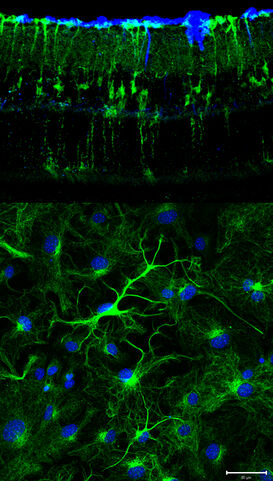 Following ON injury, we observe increased cell proliferation and nestin expression in the ciliary body, optic nerve, and neural retina. However, most of the cells known to possess stem cell characteristics and a neurogenic potential in vitro are hardly activated in vivo. Most of the proliferating cells are retinal microglia which locally divide and transiently increase in number. Surprisingly, these in situ dividing microglia ectopically express neural proteins such as nestin and NG2.
Following ON injury, we observe increased cell proliferation and nestin expression in the ciliary body, optic nerve, and neural retina. However, most of the cells known to possess stem cell characteristics and a neurogenic potential in vitro are hardly activated in vivo. Most of the proliferating cells are retinal microglia which locally divide and transiently increase in number. Surprisingly, these in situ dividing microglia ectopically express neural proteins such as nestin and NG2.
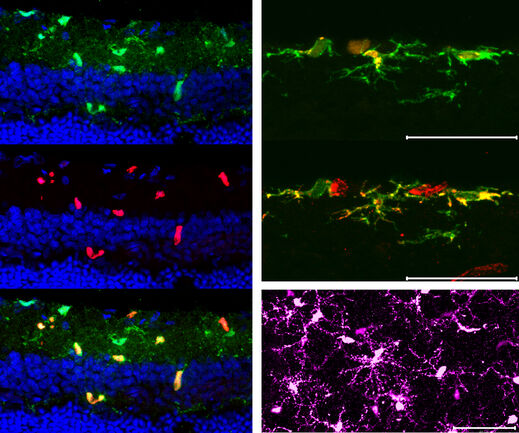
Interestingly, a subpopulation of cerebral microglia have been reported as displaying a more immature cell character, and, moreover, can give rise to neurons in vitro. In this study, we further addressed the question whether retinal microglia also possess this neurogenic potential and are able to “trans-“differentiate into neurons in vitro, and in addition, whether this potential can also be induced in vivo, following an injury.
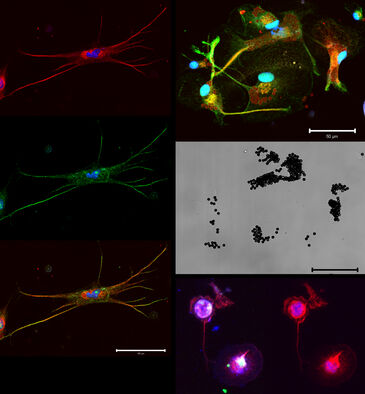
Using different transgenic mouse models, the neurogenic potential of microglia and Müller glia will be further evaluated following retinal injury, both in vivo and in vitro.
3. Analysis of neuron-glia interactions during acute retinal ischemia and optic neuropathies. Effect of treatment with erythropoietin
Neuronal injury caused by experimental lesion or disease often results in cell death. This is mainly due to an insufficient supply of trophic factors after injury. Several strategies have been evaluated to protect neurons from cell death. One of these involves the use of erythropoietin (EPO). The glycoprotein, EPO stimulates and regulates the production of red blood cells and also acts as a potent neuroprotective peptide. Neuroprotection is dependent on the presence of EPO receptors, which are mainly localized on retinal ganglion cells (RGCs) and on Müller glia in the retina. In a first approach, we evaluate the effect of an optic nerve lesion on the expression of EPO and EPOR in the retina, in particular, in glial cells. A further approach involves analysis of the glial cell response after selective treatment with EPO or its non-hematopoietic analog carbamylerythropoietin (CEPO) following acute retinal ischemia, both in vitro and in vivo. We address the question whether EPO and EPOR have a neuroprotective effect on glial and RGCs survival after injury, and whether EPO could be used as a therapeutic drug. In particular, we investigate the in vivo and in vitro effect of acute retinal ischemia and EPO or CEPO treatment on the expression of the mitochondrial enzyme frataxin, reported as being neuroprotective.